The New Zealand Government has announced the approval of weight loss drug Wegovy.
In an interview with the Herald, Associate Health Minister David Seymour (soon to be our Deputy PM) welcomed the move, but said the process of approving medicines in New Zealand remains too slow. He said the approval of Wegovy is “very exciting” and continued “it will be a game changer for a lot of people”. Seymour enthused that the drug will prevent people going to hospital and overall save our health system money.
Over in America, where weight loss drugs have become a part of the weird celebrity scene, a significantly different story is unfolding that Seymour has inexplicably missed. The results of a 2025 study of 215,000 users conducted at Washington University has been published by Nature Medicine under the title “Obesity drugs: huge study identifies new health risks“. Whilst confirming that the injectable drugs can assist weight loss and reduce the impact of a number of associated illnesses such as stroke, heart disease and diabetes, they described the risk reduction as modest. Significantly, researchers found a wide range of serious adverse effects. These included a doubled risk of pancreatitis, an 11% increase in the risk of arthritis and an elevated risk of low blood pressure and serious kidney disease.
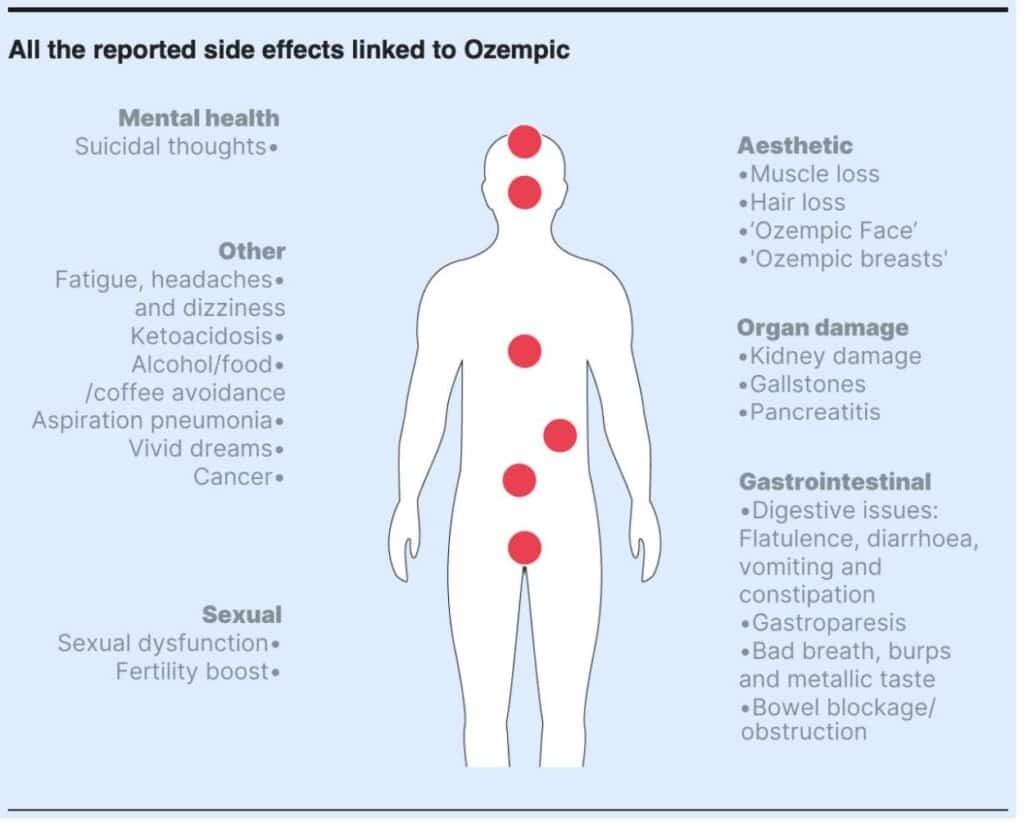
The Washington University researchers warned that the adverse effects can be “very severe” and “need to be recognised”. The full list of side effects reads like a veritable Who’s Who of disease. Contrary to Seymour’s promise that Wegovy could start to empty our hospitals, the UK Daily Mail reported in January that the number of people in hospital as a result of weight loss drug side effects has rocketed.
This appears to be another case of the long term adverse effects of novel drugs starting to kick in as their popularity and length of use increases. The UK Medicines and Healthcare products Regulatory Agency (MHRA) records that 400 people have been hospitalised with weight loss drug side effects so far with the most recent numbers soaring by 46% in one month alone.
More worrying still, a January study published in JAMA Ophthalmology is entitled “Ophthalmic Complications Associated With the Antidiabetic Drugs Semaglutide and Tirzepatide” It examines the reports of blindness following weight loss drug use. An article in the NY Post published on March 8 is entitled “We went blind after using weight-loss drugs like Ozempic and Mounjaro”. It reports that one US firm of Attorneys alone has received hundreds of GLP-1 (the main ingredient of weight loss drugs) medication and vision loss cases. A 56 year old mechanic from New Jersey told the paper: “I’ve been a mechanic my whole life but I can’t do anything with my hands anymore,” said Norris, who stopped taking Mounjaro around July 2024 but still has significantly impaired vision.
The growing prevalence of a wide range of adverse effects highlights one possible effect of the New Zealand Gene Technology Bill. The Bill mandates that drugs approved in two overseas medical jurisdictions will be automatically approved here. This raises the spectre of misleading patient information and widespread unforeseen health problems, as happened during the pandemic. Informed consent is increasingly missing in action in New Zealand as health authorities appear to be taking a “we always recommend the best drug” approach, rather than rigorously informing patients of their options and the risks.
Automatically reaching for a prescription drug along with the acceptance of adverse effects have both become normalised to the detriment of public health. Obesity is an area crying out for more considered attention. It is easy to dismiss the condition as solely the result of over eating or as some would term it ‘lack of self control’, but while these factors are highly significant and known to be controlled through lifestyle changes including exercise, diet, meditation and counselling, recent research implicates another critical factor. A 2022 study entitled “Obesity—an unexplained epidemic” published in the American Journal of Clinical Nutrition identifies industrial food production as playing a central role in the growth of obesity since the year 2000.
Approval of a weight loss drug is not a permanent remedy for obesity, it might just be another step along a route to industrial food, risky drugs and the proliferation of chronic disease. The Gene Technology Bill will deregulate the production and use of many genetically altered foods which, as we have reported here and here, are subject to high levels of contamination. Lack of provision in the Bill for testing, labelling and traceability will leave the public in the dark about what they are buying and eating and the possible effects.
There is currently a political storm in New Zealand over the repugnant social media posts of a Green MP, it is worth reflecting that the MP involved has virtually no political power or influence and therefore his worrying behaviour is unlikely to cause any lasting harm to the general population. The hullabaloo is legitimate, yet we shouldn’t lose sight of the actions of the current government whose Gene Technology Bill is set to have a lasting impact on the health and well being of the whole population and that of future generations. It is those who wield real political and regulatory power in the government whose decisions can cause widespread harm to public health, especially if they fail to complete their due diligence or keep up to date with research findings.